McGreevy, P., & Yeates, J. (2018). Horses (Equus caballus). In Companion Animal Care and Welfare. Companion Animal Care and Welfare.

Abstract: Summary Domestic horses are equid members of the class Mammalia, order Perissodactyla, and family Equidae. Horses are obligate herbivores, with nutritional requirements as listed in a table. Adequate space is necessary for exercise, exploration, flight, sharing resources, play, and rolling. Company is essential for all horses, including stallions. Company provides opportunities for mutual grooming and play and allows horses to stand head-to-tail to remove flies. Unhandled horses may respond to humans as they would to predators, whereas handled horses' responses depend on their previous interactions with humans. Horses can suffer from several diseases as listed in another table. The best method of euthanasia of horses is usually sedation followed by either cranial shooting or the injection of an overdose of pentobarbitone into the jugular vein. Behavioural signs of distress can include increased locomotory activity, vigilance behaviours, neighing, snorting, pawing, nibbling walls and buckets, defaecation, rearing, kicking stable walls or doors, and high-stepping 'prancing'.
|


|
Malavasi, R., & Huber, L. (2015). Referential communication in the domestic horse (Equus caballus): first exploration in an ungulate species. In Proceedings of the 3. International Equine Science Meeting.

Abstract: An important question in the study of animal communication is whether non-human animals are able to produce communicative gestures, i.e. nonvocal bodily actions directed to a recipient, physically ineffective but with a meaning shared in the social group [1]. Passive gestures are instrumental, tuned to the mere presence/absence of others, whereas active informers recognize receivers as communicative agents and activate shared-attention mechanisms for identifying their attentional state (SAM [2]; e.g. Schwab and Huber [3]). Six operational criteria must be evaluated to classify a signal as referential and intentional [4]: (1) alternative gazes between the partner and the target; (2) apparent attention-getting behaviours are deployed; (3) an audience is required to exhibit the behaviour; (4) the attentional status of an observer influences the propensity to exhibit behaviours; (5) communication is persistent and (6) there is elaboration of communicative behaviour when apparent attempts to manipulate the partner fail. Dogs [5] and non-human primates (reviewed in Liebal and Call [6]) can tune a human receiver’s attention to the object of interest by combining directional and attention-getting signals, such as turning the head or body, gazing to the receiver, and/or establishing eye contact. Research on other species is scarce.
Horses rely on humans to survive in domestic settings and may have evolved skills for communicating flexibly with them [7]. Horses understand human attentional cues (such as body and head orientation, eyes opened/closed) [8], permanent pointing [9] and, to some extent, gazing [10]. Here we tested the ability of 14 outdoor, herd-living domestic horses to communicate referentially with a human partner about the location of a desired target, a bucket of food out of reach. After the baiting of two buckets placed in opposite, unreachable locations were shown by the experimenter, the subject would walk to one of the two buckets. Because approaching a bucket would reveal that the food is out of reach, we expected the horse to look back to the experimenter, then to the bucket, and alternate this gazing several times to indicate its intention. To test whether our prediction is correct and alternate gazing is indeed the result of the horse's referential communication, we video-recorded the behaviour of the subjects in the test (FORWARD) and three control conditions: (1) FORWARD: experimenter oriented to the center of the arena, (2) BACK: experimenter backward oriented in respect to the arena, (3) ALONE: experimenter absent, (4) MANY: as FORWARD plus a familiar human oriented to the subject behind the bucket (Figure 1). We used a conservative criterion of back gazing by considering only turning the head back more than 90 degrees. The results confirmed our prediction. The horses alternated gazes between the partner and the buck significantly more often in the FORWARD than in all the other conditions (Table 1), thus satisfying operational criteria #1, #3 and #4. They also alternated head nods with gazes to the partner significantly more often during the FORWARD condition. We thus considered head nods not an instrumental signal of arousal, but an attention-getting behaviour with communicative function. Subjects used both head nods and neck stretched toward the buck more often in the FORWARD than in the BACK and the ALONE conditions, thus satisfying criteria #2, #3 and #4. In condition MANY, the frequency of head nods did not differ from condition FORWARD, probably because nods were directed to the additional partner behind the buck. This also satisfies criteria #4. The horses gazed to the partner most often in the FORWARD than in the BACK and the MANY conditions, but not in the ALONE. In this condition, subjects could observe the partner walking further from the test arena. To test for the different functions of gazes in presence and in absence of the partner, we compared their average duration between the two conditions: the significantly longer duration of gazes when the subject was alone suggests the instrumental monitoring function of gazes in this experimental condition.
Altogether, the findings suggest that domestic horses possess the ability to use referential communication in an interspecific context, but additional analyses are needed to test for operational criteria #5 and #6. Flexible and voluntary use of communicative signals reveal sophisticated cognitive processes involved in the strategic emission of these signals, and the finding of referential communication skills in an ungulate species forces us to reconsider the evolutionary path of intelligence. Furthermore, ungulates are used intensively by humans (transportation, meat, agriculture, leisure activities), and their welfare is often compromised. Determining whether ungulates can communicate their needs and preferences is paramount to a proper ethical management.
|

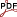

|
Baragli, P., Demuru, E., & Palagi, E. (2015). Mirror on the wall, who is the horsest of our all? Self-recognition in Equus caballus. In Proceedings of the 3. International Equine Science Meeting.

Abstract: Mirror Self-Recognition (MSR) is an extremely rare capacity in the animal kingdom that reveals the emergence of complex cognitive capacities (de Waal 2008). So far, MSR has been reported only in humans, chimpanzees (Gallup, 1970), bottlenose dolphins (Reiss and Marino, 2001) and Asian elephants (Plotnik et al, 2006), all species characterized by a highly developed cognition. There is growing evidence that domestic horses posses high cognitive abilities, such as cross-modal individual recognition (Proops et al, 2009), triadic post-conflict reunion to maintain social homeostasis (Cozzi et al, 2010), complex communicative systems (Whatan and McComb, 2014), flexibility in problem-solving (Lovrovich et al, 2015), and long-term memory (Hanggi and Ingersoll, 2009). All these capacities make horses a good candidate to test the ability of MSR in a domestic species. Through a classical MSR experimental paradigm (de Waal 2008) we tested eight horses living in social groups under semi-natural conditions (from the Italian Horse Protection rescue centre). Animals showing MSR typically go through four stages (Plotnik et al, 2006): (i) social response, (ii) physical mirror inspection (e.g., looking behind the mirror), (iii) repetitive mirror-testing behaviour (i.e., the beginning of mirror understanding), and (iv) self-directed behaviour (i.e., recognition of the mirror image as self). The final stage, known as the “mark-test”, is verified when a subject spontaneously uses the mirror to check for a coloured artificial mark on its own body which it cannot perceive otherwise. The horses underwent a three-phase “mark-test”: 1) with sham mark (transparent ultrasound water gel) positioned on both side at jaw level, 2) mark (yellow eye shadow mixed with ultrasound water gel) positioned on left side of jaw (with sham mark on the right), 3) mark (yellow eye shadow mixed with ultrasound water gel) positioned on right side of jaw (with sham mark on the left)
The mirror was one 0.5-cm-thick piece of 140x220-cm plexiglass glue on wood. Each test lasted one hour, horses were tested once a day, in consecutive days and at the same time. Our preliminary result on 1 horse shows some changes in self-directed behaviours which can be attributed to presence of the coloured mark. Firstly, the presence of the coloured mark significantly increased the frequency of scratching on both sides of the muzzle (p < 0.0001). The most intriguing result (p < 0.0001) comes from the comparison of the scratching rates directed towards the coloured mark side (N = 41) and the sham mark side (N = 23). Under the control condition (i.e. sham mark on both sides) no statistical difference was found for the scratching rates directed to the muzzle sides (dx N = 8; sx N = 5). Although further analyses are needed to confirm these preliminary results, our finding opens new scenarios about the evolution of Mirror Self-Recognition. The capacity of horses to recognize themselves in a mirror may be the outcome of an evolutionary convergence process driven by the cognitive pressures imposed by a complex social system and maintained despite thousands years of domestication.
Keywords:
Domestic horse · Mark test · Socio-cognitive skills · Self-awareness
References
De Waal FBM (2008) The thief in the mirror. PloS Biol 6(8):e201
Gallup GG Jr (1970) Chimpanzees: Self-recognition. Science 167: 86-87.
Reiss D, Marino L (2001). Mirror self-recognition in the bottlenose dolphin: A case of cognitive convergence. Proc Natl Acad Sci U S A 98:5937-5942.
Plotnik J, de Waal FBM, Reiss D (2006) Self-recognition in an Asian elephant. Proc Natl Acad Sci U S A 103: 17053-17057.
Proops L, McComb K, Reby D. (2009) Cross-modal individual recognition in domestic horses (Equus caballus). Proc Nat Acad Sci USA;106:947-951.
Cozzi A, Sighieri C, Gazzano A, Nicol CJ, Baragli P. Post-conflict friendly reunion in a permanent group of horses (Equus caballus). Behav Process 2010;85:185-190.
Wathan J, McComb K. The eyes and ears are visual indicators of attention in domestic horses. Curr Biol 2014;24(15): R677-R679.
Lovrovich P, Sighieri C, Baragli P (2015) Following human-given cues or not? Horses (Equus caballus) get smarter and change strategy in a delayed three choice task. Appl Anim Behav Sci, in press.
Hanggi EB, Ingersoll JF. (2009) Long-term memory for categories and concepts in horses (Equus caballus). Anim Cogn; 12:451-462.
|


|
Bartoš, L., Bartošová, J., Pluhácek, J., & Šindelárová, J. (2011). Promiscuous behaviour disrupts pregnancy block in domestic horse mares. Behavioral Ecology and Sociobiology, 65(8), 1567–1572.

Abstract: Based on questionnaires from horse breeders, we found that bringing a pregnant mare which had been mated away from home into a vicinity of a familiar male who was not the father of her foetus increased probability of pregnancy disruption. These mares aborted in 31% of cases, while none of those mated within the home stable aborted. Repeated sexual activity either by a stallion or dominant gelding from the normal home group was observed shortly after the mare came from away-mating. Pregnant mares isolated from home males by a fence were even seen soliciting them over the fence. We speculate that, once returned to the home “herd”, and introduced to familiar males, mares were more likely to terminate their pregnancy to save energy and avoid likely future infanticidal loss of their progeny by dominant male(s) of the home social group. This is a newly discovered phenomenon where a mare manipulates the male’s paternity assessment by promiscuous mating. It may explain a common increased incidence of foetal loss in domestic horses occurring in nearly 40% of pregnancies. We conclude that the common practice of transporting the mare for mating and then bringing her back to an environment with males, stallions or geldings, which did not sire the foetus, is the main cause of high percentages of pregnancy disruption in domestic horses.
|


|
Komárková, M., & Bartošová, J. (2013). Lateralized suckling in domestic horses (Equus caballus). Animal Cognition, 16(3), 343–349.

Abstract: Brain lateralization enables preferential processing of certain stimuli and more effective utilization of these stimuli in either the left or the right cerebral hemisphere. Horses show both motor and sensory lateralization patterns. Our aim was to determine whether a lateralized response could be detected in foals during the naturally side-biased behaviour, suckling. The foals’ preferred suckling side could be the effect of either visual or motor lateralization. In the case of a visual lateralized response, foals are expected to suck more often from the mother’s right side, so potential danger can be detected by the better adapted right hemisphere (i.e. left eye). Motor lateralization can be identified when a foal will suck predominantly from one side, either left or right. We found no population trend in the preferred suckling side, but we detected significant differences amongst individual foals. One-third (35.4 %) of 79 foals showed a strong, either right or left side preference which increased with age. The mothers did not influence the foals’ suckling side preferences either by side-biased rejection or termination of suckling. According to our findings, a general pattern of sucking with the left eye open for better danger detection and recognition is unlikely in foals up to 7 months old. Foals of this age are probably young or fully focused on suckling and rely on their mothers’ vigilance. Individual side preferences amongst foals are suggested to be based on motor lateralization.
|


|